Your teacher will show you a demonstration in which water ice is placed on two different surfaces.
The main this here is to have students discuss aloud their ideas and to expose them to the concepts. It’s important to listen to what they have to say and to validate their ideas while leading them to a better understanding of what is going on.
The measured temperature should be near or below 4°C. Mention that water freezes at 0°C.
The heavy one is made of solid aluminum. It will feel cold to the touch due to its high conductivity for heat. It is heavy and smooth.
The light one is made of a plastic foam, which can be seen on the sides. The surface is textured and it feels warm to the touch.
Both should be room temperature, about 22°C
ĀĀStudents probably expected the heavy square to be colder than the lighter one. Explain that since they are in the same room in contact with the same air they can be expected to have the same temperature.
ĀĀStudents will likely anticipate that there is some trick. Get them to predict which square’s ice cube will melt faster when they ignore the feeling that there is something funny going on. Elicit some thoughts and explanations and reflect them back.
ĀĀThe ice cube on the aluminum square should have melted completely within a minute. The one on the plastic foam should have melted hardly at all. This is because the aluminum is a good conductor while the foam is an insulator. Heat can move easily into the ice cube on the aluminum but only very slowly into the one on the foam. The point is that it is heat that drives a phase change such as melting, not temperature. Both squares had the same temperature (or nearly) but the ice cube melted faster on the square that could provide the heat faster by conducting it from the surroundings.
ĀĀThe temperature of the ice and water in the styrofoam cooler should still be about the same but the ice has in fact melted a bit: some pieces pulled out are smaller than before. The temperature does not have to rise for ice to melt. The ice just has to absorb some heat, which this ice has done by absorbing it from the air.
Heat is what melts ice and causes water to evaporate. Heat has to be added change the temperature of something to its melting point temperature but after that the heat causes the phase change and the temperature does not have to change.
The informational section has been left out of this document.
In the spaces below create some molecule drawings for the phases of carbon dioxide (CO2). A molecule of carbon dioxide has one atom of carbon in the middle bonded to two atoms of oxygen and looks like this:
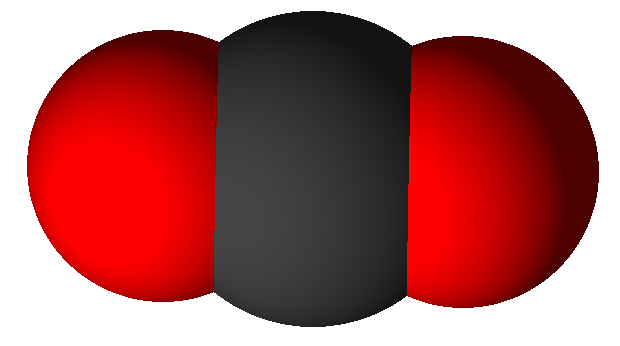
|
Ā The drawing should show multiple copies of the carbon dioxide molecule neatly arranged in a regular pattern. (tightly packed, nice and regular) Solid |
Ā |
Ā The drawing should show multiple molecules randomly arranged with a bit more space between them. (a bit looser but random) Liquid |
Ā |
Ā The drawing should show just a few molecules widely spaced apart and randomly arranged. (very few and far apart) Gas |
Deliberately leaving out the safety information because it is on the main handout.
Your teacher will show you how to do each of the demonstrations below. You will have a chance to try each of them and record your observations.
Singing SpoonHold a piece of dry ice with a pair of tongs pressed against the table top. Press a warm spoon against the dry ice.
|
Air HockeyUse tongs to place a piece of dry ice on the table. Let it sit awhile: look at it closely while you wait about a minute. Once the bottom of the piece of dry ice has flattened out, try hitting it from the side to make it slide.
|
Pop-top Film CanistersUse tongs to place a small piece of dry ice in one of the film canisters. Seal it up (this is safe because the top will pop off before the container builds up enough pressure to explode).
|
FogUse tongs to place one or two pieces of dry ice into a cup of cool water. Use tongs to place one or two pieces into a cup of very warm water.
|
Students may or may not have any further observations or questions but this could be a fun part of a post-activity discussion.
The term is sublimation. A solid sublimes when it turns directly into a gas without melting first.
The carbon dioxide never melted so we could not see it in its liquid phase. At some point the teacher may mention (now or earlier) that in order to be a liquid carbon dioxide needs to be at a very high pressure: over 5 times atmospheric pressure. Equivalent to being 53 meters (173 ft) under water.
We observed this very thing as we played with the dry ice.
Students may think not since it is difficult to observe and probably none of them ever have. Two examples that may resonate are snow banks and frosty freezers. Snow banks can diminish in size when the sun shines on them without melting. This is because the ice is subliming. Freezers get all frosty inside over time as water sublimes from food and ice cubes and then freezes again on other surfaces.
This is really an advanced question that today’s experience may not give students the necessary experience to answer properly. However, it is a good idea to plant seeds of ideas so that they question assumptions in the future.
Heat exchange is what causes phase changes. In order to overcome the weak stickiness that affects all molecules heat must be absorbed to make the molecules break away from each other. The heat that breaks those connections can’t make the temperature rise so phase changes often happen at one constant temperature. The ice melted in the first demonstration while the temperature of the aluminum went down. This is because heat is used up in melting the ice. The ice and water in the cooler at the beginning remained about the same temperature while the ice cubes melted because they absorbed heat to cause the melting and there was none left to raise the temperature.