and probability. The speed and even the location
of objects in this world are subject to uncertainty.
Electron Orbitals
Werner Heisenberg, one of the founders of Quantum Mechanics, was
the first to describe this uncertainty mathematically. The
Heisenberg Uncertainty Principle states that it is
impossible to know with high levels of certainty both the location
and the velocity of an electron. The better you know where
an electron is, the worse your knowledge of where it is going.
The practical meaning of this is that the common picture given of
atoms that looks like a solar system is not just a little inaccurate.
It is dead wrong. Because of the uncertainty principle electrons cannot orbit in nice simple circles because if they did it would be possible to know their exact position and speed at any moment. Diagrams such as the one at right are useful for
counting electrons and illustrating different energy levels. But they are
worthless as pictures of what atoms really look like.
The solar system model of the atom uses orbits to show
where the electrons are. Electrons do not really orbit the nucleus.
They do live in regions of space called orbitals .
Orbitals are organized by the energy level or shell that they belong
to.
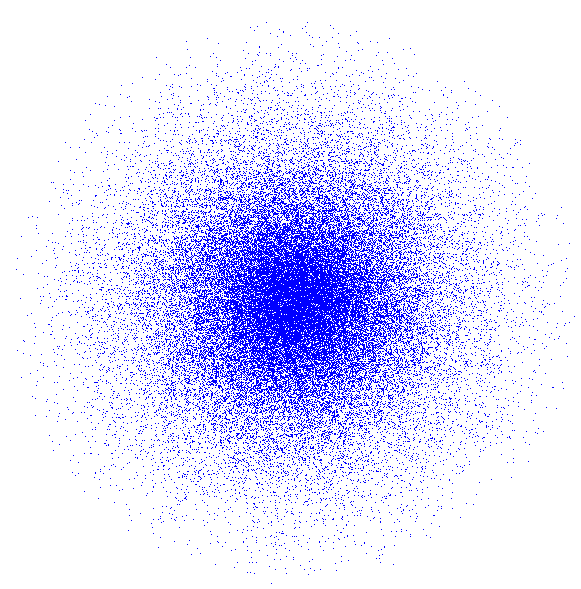 |
The 1s Orbital
|
The uncertainty principle tells us that we cannot pin down the electron’s position exactly. Quantum physics calculations allow us to calculate the probability that an electron will be in a particular location. The combination of a large number of such calculations gives rise to a diagram like the clouds pictured on this page. The quantum physics equations reveal that in the lowest energy level the electrons can be found in a spherical region around the nucleus. Other orbitals have different shapes, as we will see. An orbital is the
shape of the region in space where an electron is most
likely to be found. At left you see what is called the 1s
orbital. This ball-shaped orbital gives a better idea of what a
hydrogen atom actually looks like. The dots in the picture represent
possible positions of the one electron the hydrogen has. The nucleus
(too small to see) is in the center of the picture. Notice that there
are more dots near the center of the sphere than near the edges. This
shows that the electron is most likely to be near the nucleus. But
that does not mean it is impossible to find the electron very far
away from the nucleus.
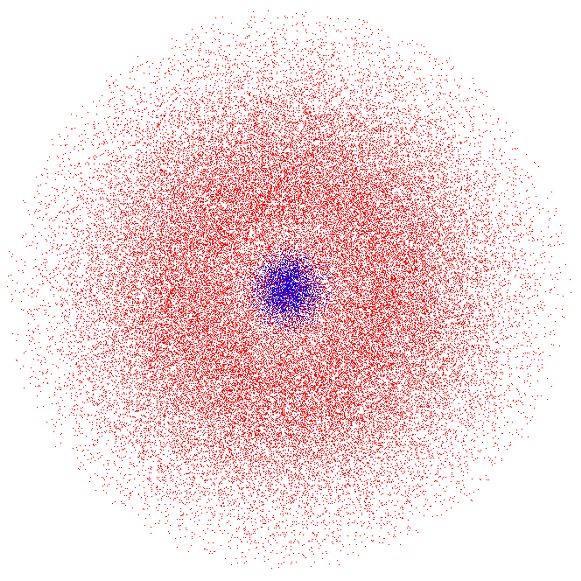 |
The 2s Orbital
|
The next larger orbital in an atom is called the 2s orbital. It is one of the orbitals found in the second energy level (or shell) of an atom. The 2s orbital of an atom is shaped just like the 1s orbital except that it is larger than the 1s
orbital. The 2s orbital is also different because it has two parts, an inner and outer sphere. An electron may be in either sphere but is never found in the border between them.
Generally, s orbitals all look alike but get bigger in higher energy levels. The same is true for the other types of orbitals: at higher energies they look similar but are bigger.
Orbitals and Energy
 |
The 2s Orbital
|
 |
The 2px Orbital
|
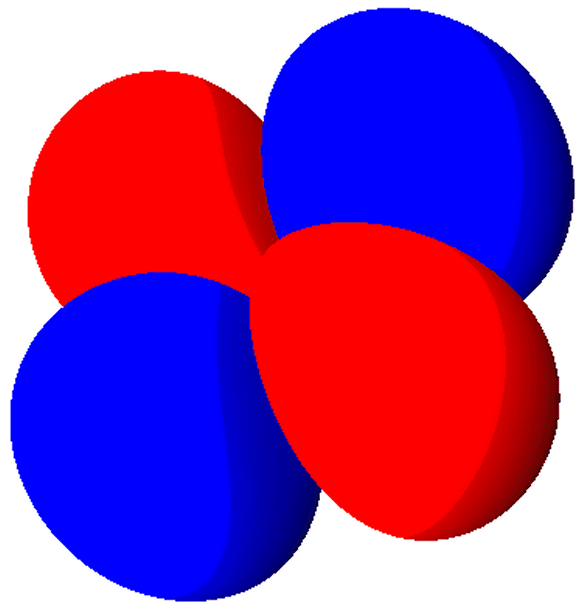 |
The 3dxy Orbital
|
Electrons in atoms are organized into energy levels or shells. In the first shell electrons have the lowest possible potential energy within the atom. In the second shell electrons have higher potential energy. The shells have different types of orbitals within them, depending on their relative potential energy. Lower shells have fewer orbitals and higher shells have more orbitals. Each orbital can have either one or two of the atom’s electrons in it.
The electrons in an atom in the ground state all have a specific location. This location is given by the shell number and by the orbital. The diagram below shows how the orbitals are organized
by their potential energy. In this activity you will learn how to
determine where the electrons in atoms and ions are placed in such a
diagram. You will also learn how to write a shorthand notation for
such diagrams.
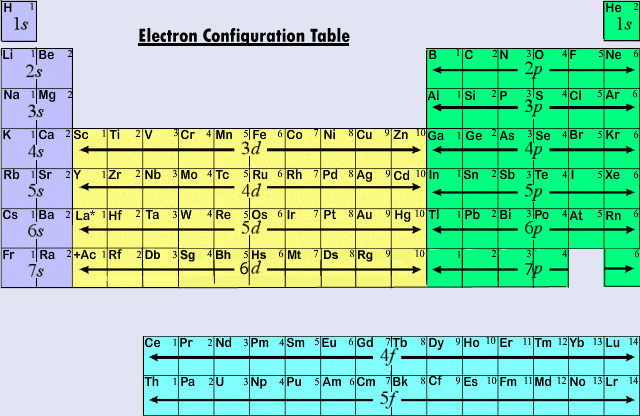
The energy levels or shells are distinguished by their number.
This number is called the principal quantum number
and is the most important quantum number for determining how much
energy an electron has. This is also called the shell number and is an integer like 1, 2, or 3.
In each energy level the orbitals have specific names. For
electrons with principal quantum number 1 there is only a
1s orbital. In the shell with principal quantum number
2 there are both a 2s orbital and three
2p orbitals. In the third shell there are one
3s, 3 3p, and 5 3d
orbitals. The pattern continues this way and at each level a new type
of orbital is added with letter-codes of f, g, h, i, etc. The original four letter-codes originate in spectroscopy in which scientists studied the lines in the emission spectrum of elements to learn about electron structure. They now do not have any relevant meaning but originally referred to features in the spectrum that were sharp, principal, diffuse, or fundamental.
Different orbitals have different shapes. The s-orbitals are all spherical and there is only one s-orbital in each
shell. The p-orbitals all look like barbells made out of balloons and
there are always three p-orbitals per shell. Each p-orbital points in a direction perpendicular to the others (px, py, pz). Most of the d-orbitals
look like various combinations of four balloons. There are always
five d-orbitals in a shell that contains d-orbitals. There are seven f-orbitals in shells which can contain f-orbitals. Only shells 4 and higher can contain f-orbitals. The shapes of f, g, and other orbitals are more complicated and are really of no importance at this point.
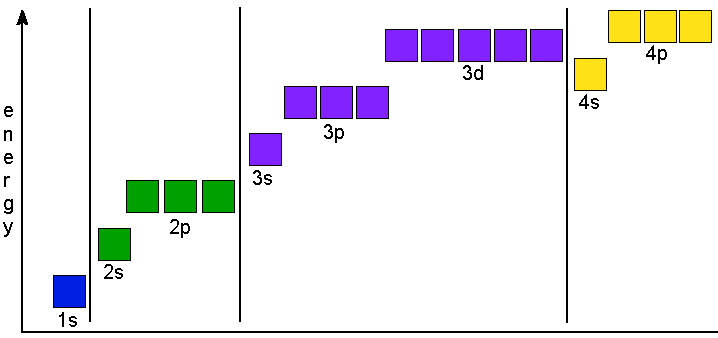
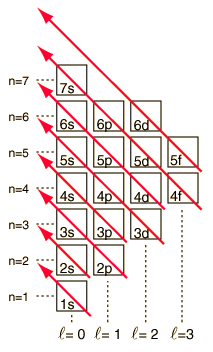
It is usually the case that each new set of orbitals is at a higher potential energy than the last set. In the diagram above all of the shell 3 orbitals are higher than the shell 2 orbitals. One exception in this diagram is the 4s orbital which is lower in energy than the set of 3d orbitals. Since electrons in an atom in the ground state fill the orbitals from the lowest to the highest energy this is an important exception to keep in mind.
page break
Filling Orbitals: the Apartment House Analogy
To figure out where all of the electrons in a ground-state atom are located we can follow the set of rules given in the table below. Next to these rules are some rules that are intended to be similar in spirit. The idea is that filling up the orbitals in the shells of an atom is like renting out apartments in a large building. That is, provided that the superintendent of the building has to follow a very strange set of rules.
| Electron Rules |
Apartment House Rules |
| Aufbau Principle: the electrons fill the
available orbitals from lowest energy to highest energy. In the
ground state all the electrons are in the lowest possible energy
level. |
From the Bottom Up : Rooms must be filled from
the ground floor up. Fill the one room on the first floor before
starting to put new tenants on the second floor. Then fill the s room
before the p rooms. At higher floors the order might change a
bit. |
| Hund’s Rule: The electrons must be placed
into the orbitals in such a way that no pairs are put together unless
absolutely necessary. That is, single electrons must be placed into
boxes first and then paired up if necessary. |
Singles First : the owner of the building wants
to have the tenants spread out as much as possible. For that reason
singles are placed in rooms before couples. If couples must be placed
into a room then all of the other rooms on that floor must already
have a single in them. |
| Pauli Exclusion Principle: Electrons come in two
varieties based on the direction they are ‘spinning’.
There is an Up spin and a Down spin. Up and Down spins are always
paired together and Up-Up or Down-Down combinations are not allowed.
No two electrons can ever be in the same place at the same time. |
Opposite Gender Only : When two people are placed
in a room they must be of opposite genders. No men may room together
and no women may room together. This is an arbitrary rule on the part
of the owners: in a just world we wouldn’t have to follow it.
But quantum mechanics has nothing to do with justice. |
The first rule makes a certain amount of sense. Just as you should fill a bookcase from the bottom up, electrons are placed into orbitals from low to high potential energy. The other two rules make a bit more sense only once electron spin is taken into account. Electrons have a quantum property called spin and they are described as either spinning up or down. Electrons that spin in opposite directions may be paired up but electrons that spin in the same direction (we call this parallel spins) may not be paired up. Electrons in an atom have the lowest potential energy when as few electrons are paired up as possible.
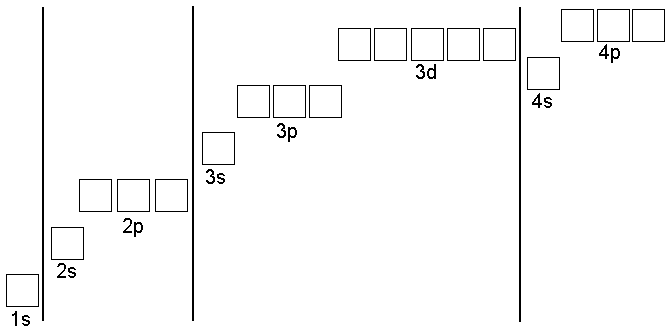
Here is an example of the rooms in the apartment house having been
filled in by the rules. The element Sulfur has 16 electrons and
starting from the bottom up you place two (1 Up and 1 Down) in the 1s
orbital.
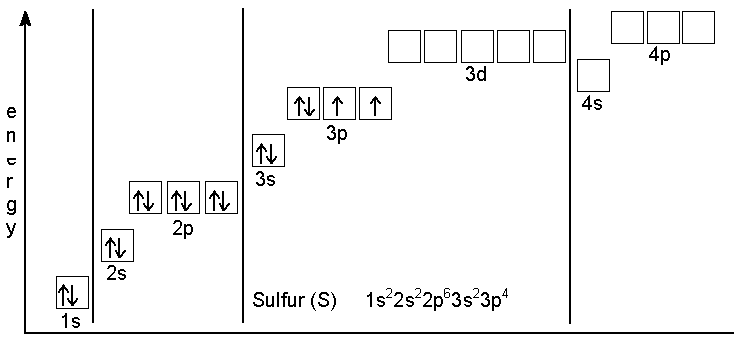
Next, electrons are placed in each of the 2p orbitals until there is one in each orbital. Then the rest of the
electrons are filled into the 2p orbitals. After that the 3s orbital
is filled and then the 3p. Electrons are placed in the boxes of the
3p orbitals one at a time until there is one in each orbital. After
that the remaining electron is placed as part of a pair in the first
3p box.
One key thing that is of great help with this is that the business
with the boxes can be abbreviated the way you see it next to the word
Sulfur in the figure at right. The short-hand gives the address of each electron and works like this:
[principal quantum number][orbital type][# of
e–]
The short-hand notation leaves off the important fact that some electrons in the outermost set of orbitals will prefer to line up with the same spin in separate orbitals. The shorthand notation for the electron configuration of Sulfur is
1s2 2s2 2p6 3s2
3p4. In that notation 1s2 means principal
quantum number 1, s-type orbital, 2 e-; 2s2
means principal quantum number 2, s-type orbital, 2 e-;
2p6 means principal quantum number 2, p-type orbital, 6
e- and so on.
page break
Connection to Valence Electrons
The valence of an element is the number that governs how many other atoms’ valences it must combine with to make a compound. It is an old-fashioned term for the ‘combining power’ of an element. Elements in the same group (the columns in the periodic table) were originally placed in those groups because they had the same or similar combining powers. You may already know that elements in the same group have the same number of valence electrons. The reason elements in the same column of the periodic table (the so-called
Groups) have similar properties is not just because they have the
same number of electrons in their valence shell. They have
similar properties because their outermost orbitals have the same
number of electrons and the same shapes. The inner shells of electrons in an atom are smaller than the outer shells. For this reason, only the outermost shell of
electrons can interact with the rest of the world. Since this shell determines the combining power of an element it is called the valence shell .
| Oxygen |
1s2 2s2 2p4 |
| Sulfur |
1s2 2s2 2p6
3s2 3p4 |
The electron configurations of Oxygen and Sulfur are given at right. Notice
that in both cases the last orbitals to fill are p orbitals. For this reason Oxygen and Sulfur belong to the p-block
of elements in the periodic table. Notice
also that there are 2 electrons in an s-orbital and 4 electrons in p-orbitals for both Oxygen and Sulfur. In elements in this block of the periodic table the valence electrons include all of those in the outermost s-orbital and p-orbital. All of the elements from
Groups 13 to 18 are part of the p-block. All of the elements
in this block fill a set of p orbitals last and all of them have
their valence electrons in s and p orbitals.
There is a rule in chemistry when it comes to understanding how elements combine. The rule is called the octet rule . The rule states that elements in the s-block and p-block of the periodic table need to have eight electrons in their valence shell in order to be chemically stable. Each s-orbital holds 2 electrons and each
p-orbital holds 6 electrons. Together they can hold the eight
electrons it takes to finish off the orbitals and make the valence
shell full.
Other sections of the periodic table are also known by the type of orbital that fills last in that section. Broadly, this way of looking at the periodic table can explain why it has just the shape that it has. In Groups 1 and 2 the s orbitals are filling. In Groups 3 through 12 the d orbitals are the last to be filled. At a principal quantum number of one (n = 1) there is only an s orbital. At n = 2 there are both s- and p-orbitals available. At n = 3 there are s-, p-, and d-orbitals available. There are no transition metals between Magnesium and Aluminum because the 3d orbitals are higher in energy, and fill up after, the 3p orbitals. Notice that the s-block always precedes the d-block and that the fourth row of the periodic table is where the first d-block elements are found. This is because the 4s orbital is actually slightly lower in energy than the 3d orbitals. (See the diagram on the second page of this packet.) Because they are lower in energy, the 4s, 5s, and 6s orbitals always fill up before the d-orbitals of the previous principal quantum number. In this activity there will not be much concern with any orbitals higher than the 4s orbital since there are some odd exceptions to the orbital-filling order that are beyond the scope of this activity.